Current Projects
Electrochemical Detection of Small Concentrations of Metal Ions inside a Microfluidic Device by Stripping Voltammetric Analysis (SVA)
Team Members
Tobias Süss, Todd Duncombe, Karl F. Böhringer
Perry Cheung, Daniel Schwartz (Chemical Engineering)
Research
Stripping voltammetric analysis (SVA) is an electrochemical method for the detection of trace metal ions in an electrolyte solution. Due to its low detection limit and high sensitivity, it is the technique of choice for the analysis of the quality of water samples. The ability to identify very small concentrations of impurities however offers a number of other applications as for example in medical diagnosis. One approach used for the detection of diseases in blood samples is to tag antibodies with a specific metal oxide nanoparticle (also called nano-barcoding). After hybridization of the antibody with an antigen (immunoassay) the nanoparticles are dissolved and the formed metal ions are detected by SVA. This allows for the identification and quantification of the antigens present in the analyte blood sample.
The analysis of immunoassays is commonly performed ex situ (Figure 1, left) and requires several mixing and washing steps. My project aims towards the integration of SVA into a microfluidic device (Figure 2, right). By restricting all the processes to a single microchannel, the loss and need of limited analyte and reagent can be minimized. In addition, the fabrication cost and power consumption can be reduced.

Figure 1: Ex situ electrochemical setup: 25ml of an aqueous solution (0.05M Cu2+, 1M KCl and 0.5M H2SO4) serves as electrolyte. Immersed into the electrolyte are the reference electrode (RE, saturated calomel electrode), the working electrode (WE, a gold thin film on a piece of silicon) and the counter electrode (CE, a carbon rod).
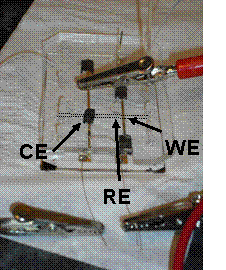
Figure 2: 1st protoype of a microfluidic device with integrated electrodes. The electrodes materials are gold (WE), carbon paste screen printed on gold (CE) and a silver wire (RE). The dashed lines indicate the channel.
